Why Flanax?
Share on:

The duration of action of a drug is known as its half life. This is the period of time required for the concentration or amount of drug in the body to be reduced by one-half. In general, a drug is no longer considered pharmacologically active after one half life. This means that a drug looses much of its activity or effectiveness after one half life.
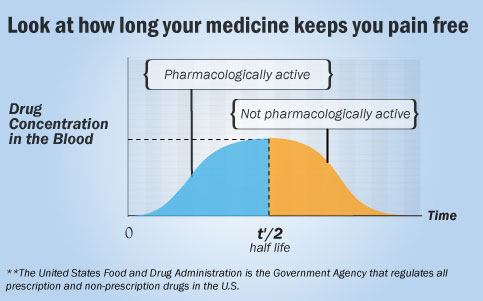
In a medical context, half-life may also describe the time it takes for the blood concentration of a drug in the human body to halve its steady-state concentration within the blood. Drug half-life is an important pharmacokinetic parameter that most doctors and pharmacists use to measure drug activity in the human body. A drug half-life is usually denoted by the abbreviation t½ in most medical literature. A drug half life is required to be reported to the United States Food and Drug Administration, by a drug manufacturer for each drug it produces in the U.S.
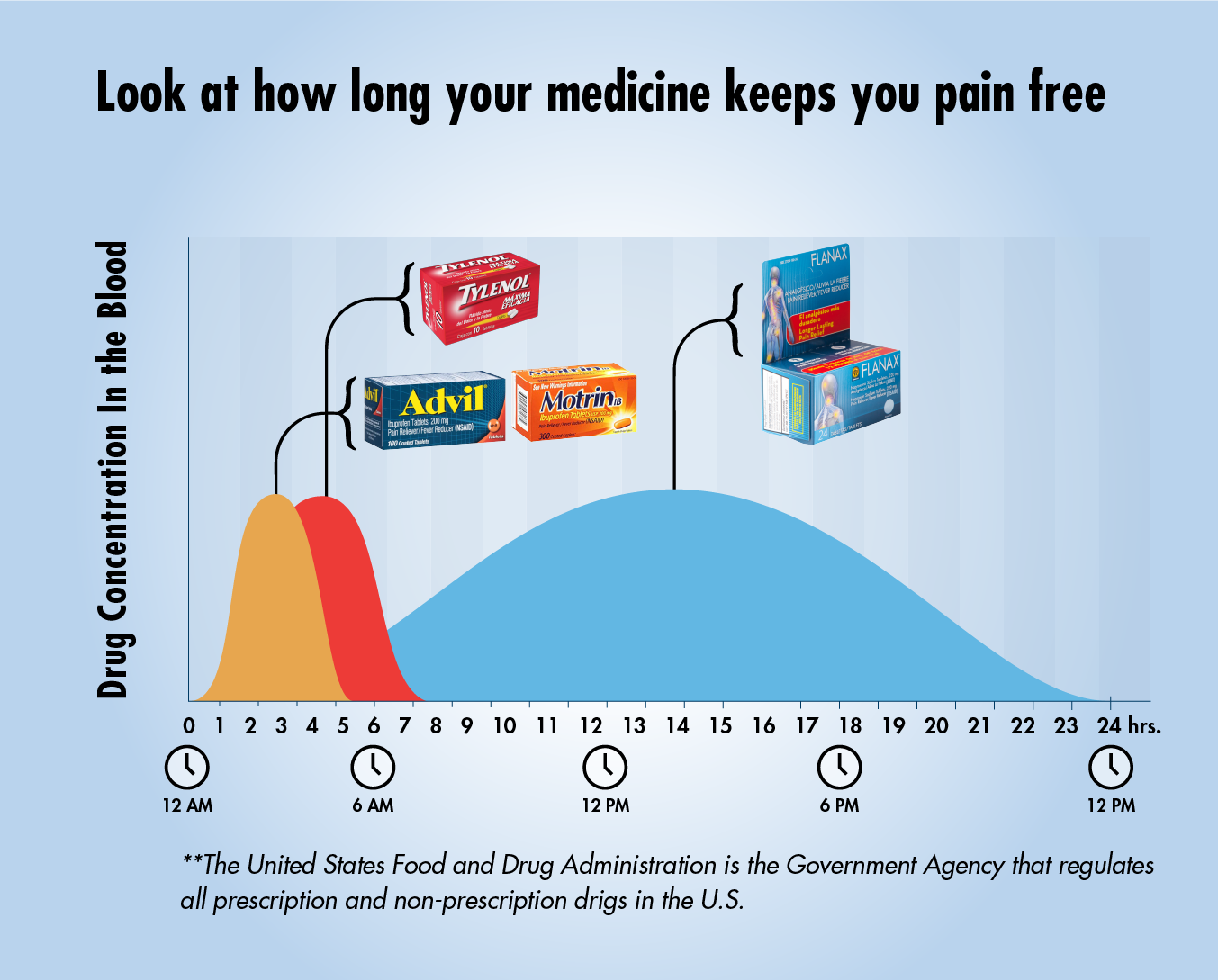
The United States Food and Drug Administration is the Government Agency that regulates all prescription and non-prescription drugs in the U.S. What is the t½ of your analgesic?
Active Ingredients
Naproxen sodium 220 mg (naproxen 200mg) as pain reliever and fever reducer.
Other information
Each tablet contains: sodium 20 mg, store at 20-25°C (68-77°F). Avoid high humidity and excessive heat above 40°C (104°F).
Inactive ingredients
- FD&C blue no. 2
- Aluminum lake
- Hypromellose
- Magnesium stearate
- Microcrystalline cellulose
- Polyethylene glycol
- Povidone
- Talc
- Titanium dioxide
